Low-energy Control of Atrial Fibrillation
Atrial fibrillation (AF) is the most common sustained cardiac arrhythmia worldwide affecting an estimated 5.5 million people worldwide. Complications associated with chronic AF include increased risk of both thromboembolism and stroke. Left untreated, paroxysmal AF often progresses to permanent AF, which is resistant to therapy. Although underlying anatomic or pathophysiological factors may fuel its progression, AF itself may lead to its own perpetuation through electric, structural, and metabolic remodeling of atrial tissue. The realization that AF begets AF has led to management strategies that are designed to avoid the progression of AF by reducing the frequency and duration of AF episodes.
One such strategy, cardioversion, attempts to reset all electrical activity in the atria and requires the use of large (>5 V/cm) electric field gradients. These high energies cause pain and trauma to the patient, damage the myocardium, and reduce battery life in implanted devices. Another strategy, antitachycardia pacing (ATP), seeks to avoid the development of permanent AF by suppressing paroxysmal AF. ATP consists of a train of 8 to 10 lowenergy stimuli delivered as a pacing ramp or burst at 50 Hz via a single pacing electrode. ATP is effective in treating spontaneous atrial tachyarrhythmias, especially slower tachycardias, but it is not very effective for converting AF.
To overcome the limitations of ATP and cardioversion, we proposed a new technique that employs a series of low-amplitude pulsed electric fields to overdrive AF and thereby terminate it, a strategy suggested by previous experimental studies with the use of physically implanted electrodes. The technique, which we call low-energy anti-fibrillation pacing (LEAP), takes advantage of the fact that after an electric field pulse, virtual electrodes may arise at interfaces separating regions with different conductivities [1]. These sites may be macroscopic, such as blood vessels or ischemic regions, or smaller-scale discontinuities, including areas of fibrosis or abrupt changes in fiber direction. Virtual electrodes may become a secondary source (i.e., a site of wave emission), depending on the extent of the conductivity discontinuity and the electric field strength.
Figure 1
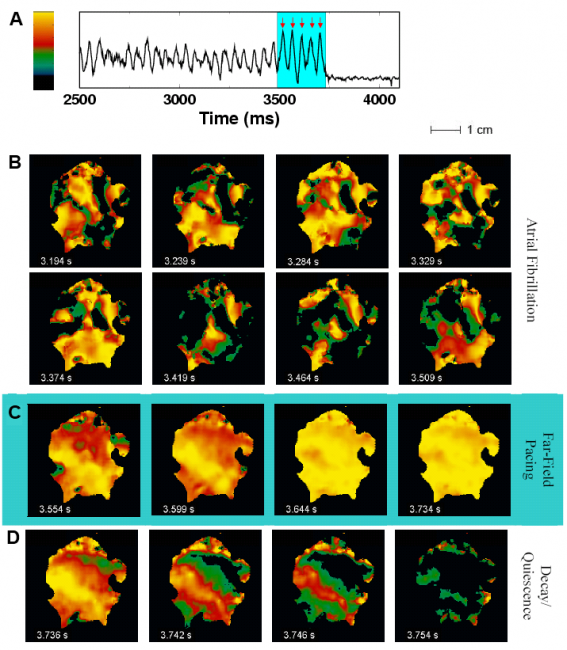
Successful termination of AF.
(A) Optical signal from 1 pixel during AF showing successful defibrillation after delivery of 5 far-field pulses of 1.4 V/cm at a cycle length of 45 ms (red arrows).
(B) Optical signal during fibrillation; color represents voltage (see color bar in (A)). Frames are 45 ms apart in time and show the complex wave patterns during fibrillation.
(C) Effects of shocks 1, 2, 3, and 5 (left to right), with the first 2 panels showing partial capture and the last 2 showing global capture.
(D) Evolution to full repolarization and quiescence after shock 5. Throughout, light blue shading indicates time during applied shocks (from [2]).
Using optical mapping in isolated perfused canine atrial preparations (Fig. 1), we have shown that a series of pulses at low field strength (0.9 to 1.4 V/cm) is sufficient to entrain and subsequently extinguish AF with a success rate of 93% (69 of 74 trials in 8 preparations) [2]. We further demonstrated that the mechanism behind LEAP success is the generation of wave emission sites within the tissue by the applied electric field, which entrains the tissue as the field is pulsed (Fig. 2).
Figure 2
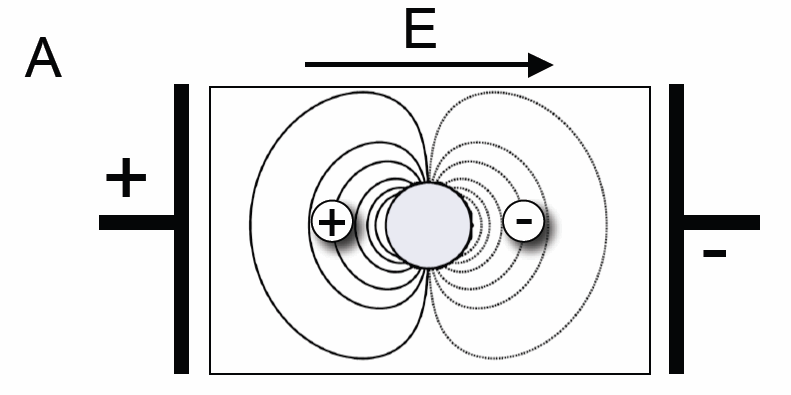
(A) Schematic of the mechanism underlying wave emission from heterogeneities in electrical conductance. In the presence of an electric field, the membrane potential in the vicinity of a heterogeneity (gray circle) will be decreased (hyperpolarized, gray lines) or increased (depolarized, black lines), with respect to the resting membrane potential. If tissue depolarization exceeds the excitation threshold in a sufficiently large region, an excitation wave will be emitted (panel B, inset).
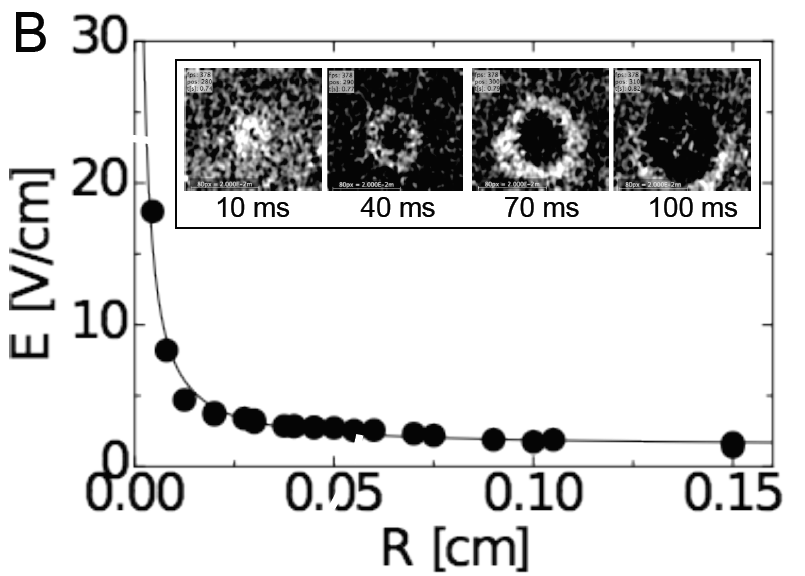
(B) Dependence of electric field strength required to induce the emission of an excitation wave from a heterogeneity of radius R. Black circles indicate data from cell culture experiments. Inset shows an example of a wave emitted from an artificial heterogeneity due to a single electric field pulse (field of view 2 x 2 cm2). Times are given with respect to the onset of the pulse.
In conclusion, we found that AF in our in vitro experiment can be terminated by LEAP with only 13% of the energy required for cardioversion. These results for AF have been confirmed in in vivo experiments. Furthermore, LEAP has been successfully applied for the low-energy termination of ventricular fibrillation in vitro and in vivo [3].
References
- A. Pumir et al., Phys. Rev. Lett. 99, 208101 (2007).
- F. H. Fenton et al., Circulation 120, 467-476 (2009).
- S. Luther et al., Nature 475, 235-239 (2011).