High-resolution Cardiac Imaging
Physiological cardiac modeling requires detailed structural, functional, and dynamical characterization of the heart. The MPRG Biomedical Physics develops high-resolution fluorescence imaging techniques (optical mapping) for intact, Langendorff-perfused hearts (see Fig. 1). Techniques from computer vision research are applied to reconstruct the three-dimensional shape of the heart from multiple silhouettes. We combine optical mapping with motion tracking, which permits, for the first time, fluorescence imaging of contractile, moving cardiac tissue. This unique experimental technique enables the simultaneous measurement of membrane voltage, intracellular calcium, and surface strain. The dynamical characterization of cardiac tissue is based on the restitution relations of action potential duration and conduction velocity, obtained from optical mapping. This functional data is complemented by structural information, including coronary vasculature and fiber orientation obtained from micro-computed tomography, diffusion-tensor magnetic resonance imaging, and histological data. Measurements on cellular and subcellular level, including patch clamp, sharp electrode patch, and STED microscopy, are available through our research partners and complement the experimental techniques developed by the MPRG Biomedical Physics.
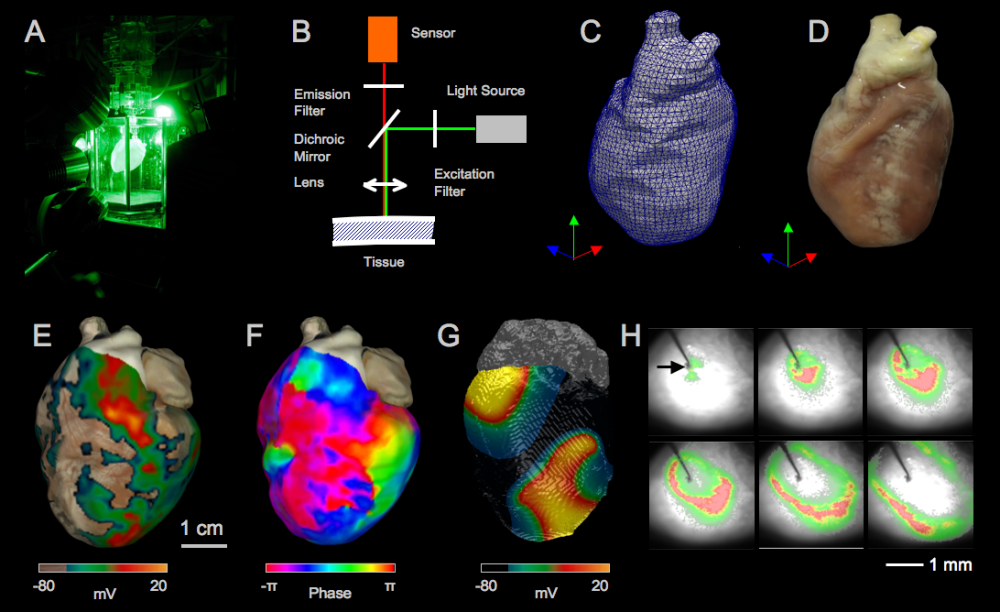
Figure 1 (A) High-resolution optical mapping of intact, Langendorff-perfused rabbit heart. (B) Schematic of the imaging setup. (C,D) Reconstructed 3D surface of a rabbit heart (C) with photorealistic texture mapping (D). (E) Complex spatio-temporal wave dynamics during ventricular fibrillation (VF). (F) Phase map during VF indicating phase singularities. (G) Numerical simulation of VF using the Fenton-Karma model [1] adapted for rabbit ventricle. (H) Pacing experiment on mouse ventricle is used to obtain action potential and conduction velocity restitutions under physiological and pathological conditions [2].
References
- F.H. Fenton and A. Karma, Chaos 8, 20-47 (1998).
- S. Petitprez et al., Circ. Research 108, 294-304 (2011).